 The bromide ion is a central nervous system depressant and chronic exposure produces neuronal effects. ( 11)īromine vapour causes irritation and direct damage to the mucous membranes. ( 7)Īntimony tribromide can be added to polymers such as polyethylene as a fire retardant. No indication of carcinogenicity (not listed by IARC). Some of the fecal antimony may represent unabsorbed antimony that is cleared from the lung via mucociliary action into the esophagus to the gastrointestinal tract. Antimony is excreted via the urine and feces. It is not known if these interactions are toxicologically significant. Antimony can covalently interact with sulfhydryl groups and phosphate, as well as numerous reversible binding interactions with endogenous ligands (e.g., proteins). Antimony is a metal and, therefore, does not undergo catabolism. Blood is the main vehicle for the transport of absorbed antimony to various tissue compartments of the body. The adrenal glands, lung, large intestine, trachea, cerebellum, and kidneys also contain relatively high levels of antimony. The hair and skin contain the highest levels of antimony. Antimony is widely distributed throughout the body. Due to its reactivity, bromine quickly forms bromide and may be deposited in the tissues, displacing other halogens. ( 3, 12, 1, 9, 10, 2)īromine is mainly absorbed via inhalation, but may also enter the body through dermal contact. Moreover, the The cause of death is believed to be essentially the same as that in acute arsenic poisoning. The antidotal action of BAL depends on its ability to prevent or break the union between antimony and vital enzymes. 
However, some studies suggest that antimony combines with sulfhydryl groups including those in several enzymes important for tissue respiration. The mechanism of action of antimony remains unclear. 
It is possible that antimony affects circulating glucose by interfering with enzymes of the glycogenolysis and gluconeogenesis pathways. The inhalation data suggest that the myocardium is a target of antimony toxicity. This is believed to be a result of bromide ions substituting for chloride ions in the in actions of neurotransmitters and transport systems, thus affecting numerous synaptic processes. The bromide ion is also known to affect the central nervous system, causing bromism. In addition, the formation of hydrobromic and bromic acids will result in secondary irritation. These free radicals are also potent oxidizers and produce tissue damage. Oral ( 9) inhalation ( 9) dermal ( 9)īromine is a powerful oxidizing agent and is able to release oxygen free radicals from the water in mucous membranes. Predicted LC-MS/MS Spectrum - 40V, Negative Predicted LC-MS/MS Spectrum - 20V, Negative Predicted LC-MS/MS Spectrum - 10V, Negative Predicted LC-MS/MS Spectrum - 40V, Positive 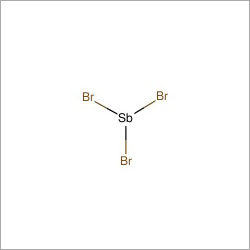
Predicted LC-MS/MS Spectrum - 20V, Positive Predicted LC-MS/MS Spectrum - 10V, Positive These are inorganic compounds in which the largest halogen atom is Bromine, and the heaviest metal atom a metalloid. Belongs to the class of inorganic compounds known as metalloid bromides. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
